|
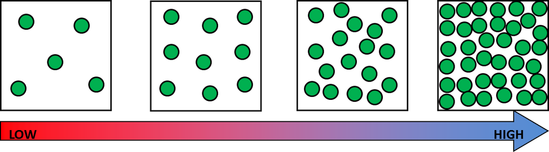
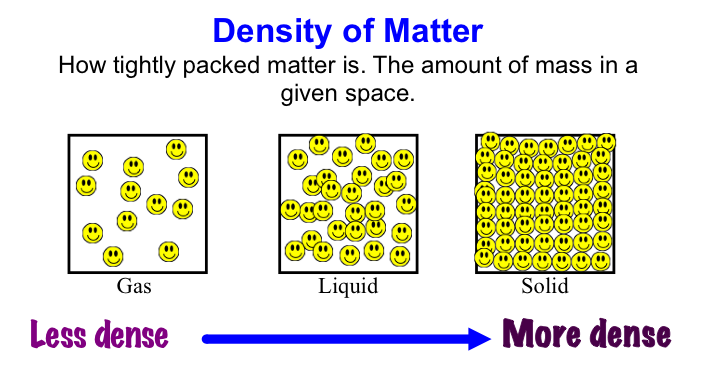
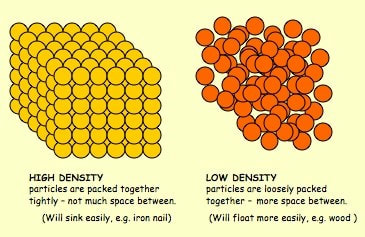
Before break, we made density towers to talk about the difference between density, mass, and volume. Here are our towers and some photos of the activity. We've done a couple of demonstrations in class the last couple of days to discuss the difference between volume, mass, and density. First we reviewed what "matter" is- anything that has mass and volume. Some people think that matter makes up "everything" but that is not accurate because matter does not make up thoughts, feelings, ideas, actions, etc. It DOES, however, make up every object. Next we reviewed the term "volume" as being the amount of space something takes up. One helpful activity is to compare three different balls (all the same volume) and answer the following question: How do the masses and volumes of a baseball, tennis ball, and mini basketball differ from each other? The balls have the same volume- they take up the same amount of space. When you use a balance scale to find the mass of these 3 balls, their masses are very different. Our results looked like this: Basketball rubber ball- 18g Tennis ball- 58 g Baseball- 148g Then we discussed the differences between each ball's mass versus its volume. The baseball had a lot more mass in the same amount of space than the other two balls. This means that it has a higher density, its molecules are packed much more tightly together. We will learn more about molecules later in the unit. Here are some graphic representations of density. As for floating (buoyancy), it isn't just how "heavy" something is that determines if it will sink or float, but a lot of it has to do with density. This explanation on density and buoyancy from http://www.indypl.org/kids/blog/?p=9042, the The Indianapolis Library's website, is excellent: Every object on earth is made of atoms. Gravity pulls these atoms to the earth. You can measure the pull of gravity on an object – we call that measurement weight. Density is how close together the molecules of a substance are or how much mass a substance has in a given space. If you have one cup of jelly beans and one cup of marshmallows…the jelly beans have more mass…there is more “stuff” compacted into the cup. The marshmallows are mostly air. If you put each of those cups in a microwave to melt…the sugar and water that makes up the jelly beans would almost fill the cup to the top. The sugar and water that makes up the marshmallows would only fill the cup a little bit because marshmallows have less mass, they are mostly made of air. Materials with more density weigh more. A cup of jelly beans weighs more than a cup of marshmallows. For an object to be bouyant, or float, it must have less density that what it is floating in, or it has to have something attached to it that helps it float – like you with a life jacket on. Here is another image of a density tower I made. As you can see, a ping pong ball, wooden cube, and foam cube all floated, meaning their density was lower than the density of the rubbing alcohol. The die, screw, and penny all sunk to the bottom, meaning they had a high density, at least equal or higher to that of the honey at the bottom.
This explanation from stevespanglerscience.com is an excellent description of all of the learning possible in this experiment. "Density is basically how much “stuff” is packed into a particular volume. It’s a comparison between an object’s mass and its volume. Remember the all-important equation: density = mass ÷ volume. Based on this equation, if the weight (or mass) of something increases but the volume stays the same, then density goes up. Likewise, if the mass decreases but the volume stays the same, then density goes down. Lighter liquids (like water or vegetable oil) are less dense than heavier liquids (like honey or corn syrup) so they float on top of the heavier liquids. The same amount of two different liquids you used in the container will have different densities because they have different masses. The liquids that weigh more (a higher density) will sink below the liquids that weigh less (a lower density)." -https://www.stevespanglerscience.com/lab/experiments/density-tower-magic-with-science/ Some of the items appeared to get "stuck" in a layer. If an object comes to rest in a certain layer, it actually means that it is less dense than the layer below it and more dense than those above it. Well, that's all for now. Hope this post helped!
1 Comment
9/17/2020 01:10:11 pm
I loved the density tower you made. It really showed the difference between the density of the liquids and solids:).
Reply
Leave a Reply. |
Archives
June 2019
Categories
All
|









 RSS Feed
RSS Feed