|
On Friday we went on our field trip to listen to the middle and high school students play instruments. It was super exciting because we got to hear science at work! The shorter instruments had higher pitches because they had a higher frequency than the longer ones, which had a low pitch and low frequency. Okay, maybe I am the only one who found this exciting, but the concert was great :) Here are some videos of one of our friends doing a demonstration- not an easy task, but she did great!
0 Comments
So.... yes. The vocabulary for this unit can be VERY confusing :) Last week in class, we came up with a way to relate the concepts back to the kids in the form of an analogy. Here it goes!
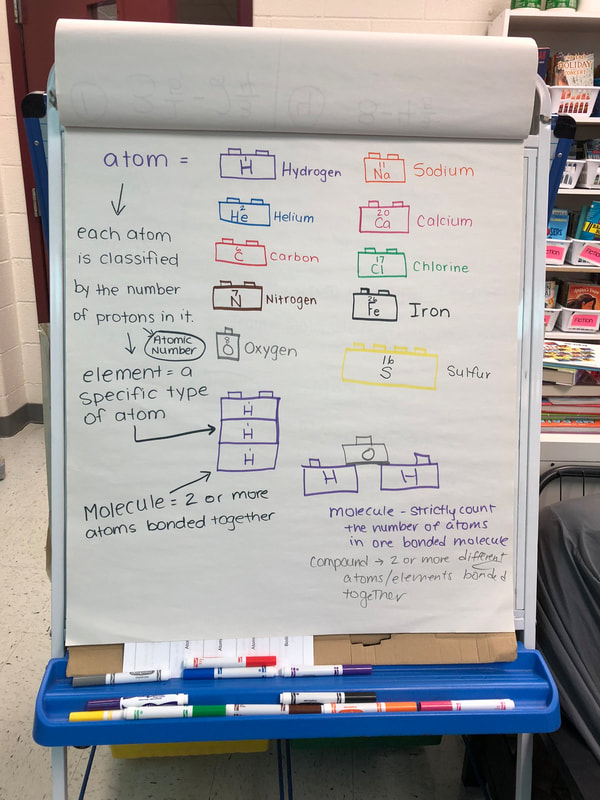
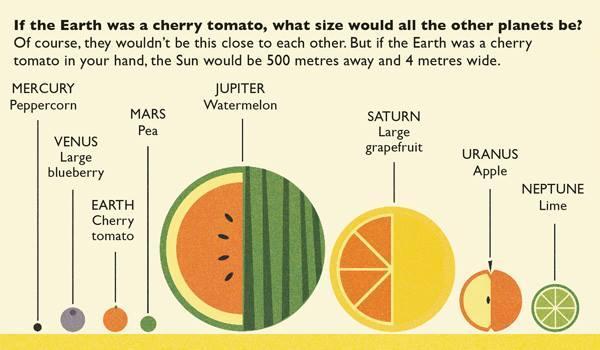
Let's say we have an Algonkian basketball league player. This player wears a yellow jersey because he is a member of the team Lightning Bolts. Now, there is another player, and she is wearing a green jersey. She wears a black jersey because she is a member of the team Random Guys. Each player that is a "Random Guys" has a black shirt, and each player that is a "Lightning Bolt" has a yellow shirt. There are five Lightning Bolt players that stick together and play as a team. There are five Random Guys players that stick together and play as a team. Now, if you were to walk into the gym during a game, you would be able to easily tell each team's players apart from other teams' players because they all have a distinct color shirt. There are other teams, such as the green shirted "Tropical Mangoes." One day, the yellow Lightning Bolts have a player out sick. They need someone to fill in on their team, so a green-shirted Tropical Mango offers to play. They combine to make a team, and even though there are two different colored jerseys/two team types playing on the same team, they play. Okay... so what does this have to do with our vocabulary? Look below for an explanation. Let's say we have an Algonkian basketball league player. The Algonkian basketball league player is an atom. All players represent atoms in this case because an atom is a single unit of matter. This player wears a yellow jersey because he is a member of the team Lightning Bolts. Just like we can tell the type of players apart by their jersey color, we can tell an atom apart from others by how many protons it has in its nucleus. The jersey color yellow says it is a specific type of player, a Lightning Bolt; the number of protons in the nucleus tell us that the atom is a specific type of element. For example, a Hydrogen atom has 1 proton in its nucleus. Hydrogen is an element; a yellow shirt is a specific kind of player. We know if we see an atom with 1 proton in the nucleus it is a Hydrogen element atom, just like we know that a yellow shirted player is a Lightning Bolt. Now, there is another player, and she is wearing a black jersey. She wears a black jersey because she is a member of the team Random Guys. This player also represents an atom, but a different type of element (because she has a black shirt, not yellow). This atom, let's say, has 2 protons in its nucleus and therefore is an atom of the element Helium. We can tell it apart from Hydrogen (1 proton) because it has 2 protons. We know that if we see an atom with 2 protons in its nucleus it is an atom of the element Helium, just like we know that if we see a black-shirted player it is a player from the team Random Guy. Each player that is a "Random Guys" has a black shirt, and each player that is a "Lightning Bolt" has a yellow shirt. Each Helium atom has 2 protons in its nucleus, each Hydrogen atom has 1 proton in its nucleus. There are five Lightning Bolt players that stick together and play as a team. There are five hydrogen atoms that bond together to make a molecule. A molecule is 2 or more atoms bonded together. There are five Random Guys players that stick together and play as a team. There are five helium atoms bonded together to make a molecule. Now, if you were to walk into the gym during a game, you would be able to easily tell each team's players apart from other teams' players because they all have a distinct color shirt. We can tell each atom apart by the number of protons in its nucleus. There are other teams, such as the green shirted "Tropical Mangoes." There are other elements with more protons in their nuclei. Let's say that the green shirted Tropical Mangoes are like the element Carbon, which has 6 protons in its nucleus. One day, the yellow Lightning Bolts have a player out sick. They need someone to fill in on their team, so a green-shirted Tropical Mango offers to play. They combine to make a team, and even though there are two different colored jerseys/two team types playing on the same team, they play. When two or more players unite, they form a team. When two or more atoms combine, they form a molecule. Molecules can have all atom from the same element (team type) or can be a special kind of molecule with two or more elements bonded together (like our two types of players bonding together and making a team). This is called a COMPOUND. When you have a molecule with two or more types of elements' atoms bonded together, it is a compound. So, a compound is a specific kind of molecule. I know this is a lot to take in, but the more we talk about it, the more sense it will make :) So, to break down our analogy: player=atom There are lots of different types of players, like there are lots of different types of atoms. Jersey color=element The jersey color tells us what team a player is on; the protons tell us what kind of element an atom is. Team of two or more players=molecule of two or more atoms Team with two or more different colored jerseys= molecule (because it has 2 or more atoms bonded together) AND compound (because there is more than one type of element's atoms in the molecule) Last week I made a HUGE mess in class in the name of science. I did a demonstration to get the kids thinking about mass and gas. Since a lot of people assume that gases have no mass, I wanted to challenge their thought processes. Here is a video of my demo from last year as I didn't get a chance to record it this year :( After the demonstration, I explained how the it worked. Contrary to popular belief, gases DO have mass and when I added the baking soda to the vinegar, a chemical reaction happened that produced the gas carbon dioxide. Carbon dioxide has more mass than the mixture of gases (approximately 78% nitrogen, 21% oxygen, and small amounts of argon, methane, and even carbon dioxide) we breathe. As I poured the invisible gas into the bag, it caused the balance scale to tip to the side that had the gas with the higher mass and density (as it was more concentrated as it was poured out of the pitcher than the spread out molecules in the air). After discussing the scale activity, we reviewed some very important vocabulary: Matter- anything that has mass and takes up space Mass- the amount of matter in an object; in science we measure in kilograms, grams, etc. Volume- the amount of space an object takes up Density- the amount of mass packed into a given space (volume) Weight- a measure of the gravitational pull on an object; a force we measure in Newtons We also talked about the difference between weight and mass. Mass is the amount of matter in an object, and weight is the measure of the gravitational pull on the mass in an object. When talking about weight, we reviewed that gravity is a force that pulls objects toward one another. We discussed how we are pulled toward the center of Earth which is why we don't go flying off into space. We also talked about how the sun's gravitational pull keeps all of the planets in orbit around it. We also talked about how moons orbit their planets due to each planet's gravitational pull. We then had a brief refresher of fourth grade science and discussed the planets and their order from the sun. We discussed mnemonics to help us remember the order of the planets: My (Mercury) Very (Venus) Educated (Earth) Mother (Mars) Just (Jupiter) Served (Saturn) Us (Uranus) Nachos (Neptune). Another fourth grade standard is the comparison of the planets according to size, We can do this by comparing them to food :) We talked about how our weight would be different if we were to go to another planet, or even the moon. We'd still have the same MASS (all of our body would have the same amount of stuff in it), but the different gravitational pulls of the planets would have different effects on our mass, thus making us weight different amounts on each planet.
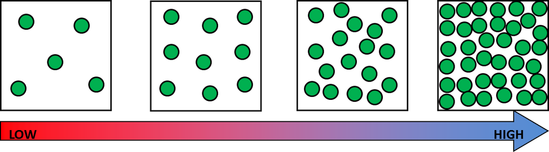
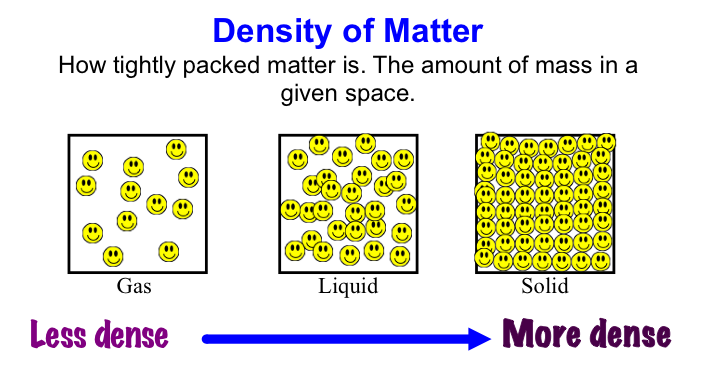
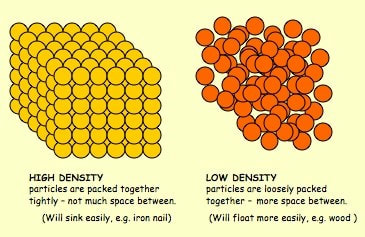
For example, if a child with a weight of 30kg on Earth were weighed on Mars (which has a gravitational pull that is only about 2/5 of Earth's), they would only weigh 11.3 kg on Mars. They still have the same amount of stuff (mass) in them, their mass just isn't being pulled toward the center of Mars as much as it is pulled toward the center of Earth. On Jupiter, that same 30kg child would way 75.8kg! The amount of gravitational pull a planet or other object has depends on its mass- the larger the object's mass, the stronger its gravitational pull. After these discussions, worked on calculating our weights on the different planets and Earth's moon. Here is a website that will let you do the same thing! Very cool! https://www.exploratorium.edu/ronh/weight/ I was having some encoding issues with these videos but have finally succeeded in uploading them. Enjoy! For shared reading last week we read and memorized the poem of the month for poetry masters. Then my little lovebugs "performed" (or screamed lol) the poem as a rap. Here is the video: Before break, we made density towers to talk about the difference between density, mass, and volume. Here are our towers and some photos of the activity. We've done a couple of demonstrations in class the last couple of days to discuss the difference between volume, mass, and density. First we reviewed what "matter" is- anything that has mass and volume. Some people think that matter makes up "everything" but that is not accurate because matter does not make up thoughts, feelings, ideas, actions, etc. It DOES, however, make up every object. Next we reviewed the term "volume" as being the amount of space something takes up. One helpful activity is to compare three different balls (all the same volume) and answer the following question: How do the masses and volumes of a baseball, tennis ball, and mini basketball differ from each other? The balls have the same volume- they take up the same amount of space. When you use a balance scale to find the mass of these 3 balls, their masses are very different. Our results looked like this: Basketball rubber ball- 18g Tennis ball- 58 g Baseball- 148g Then we discussed the differences between each ball's mass versus its volume. The baseball had a lot more mass in the same amount of space than the other two balls. This means that it has a higher density, its molecules are packed much more tightly together. We will learn more about molecules later in the unit. Here are some graphic representations of density. As for floating (buoyancy), it isn't just how "heavy" something is that determines if it will sink or float, but a lot of it has to do with density. This explanation on density and buoyancy from http://www.indypl.org/kids/blog/?p=9042, the The Indianapolis Library's website, is excellent: Every object on earth is made of atoms. Gravity pulls these atoms to the earth. You can measure the pull of gravity on an object – we call that measurement weight. Density is how close together the molecules of a substance are or how much mass a substance has in a given space. If you have one cup of jelly beans and one cup of marshmallows…the jelly beans have more mass…there is more “stuff” compacted into the cup. The marshmallows are mostly air. If you put each of those cups in a microwave to melt…the sugar and water that makes up the jelly beans would almost fill the cup to the top. The sugar and water that makes up the marshmallows would only fill the cup a little bit because marshmallows have less mass, they are mostly made of air. Materials with more density weigh more. A cup of jelly beans weighs more than a cup of marshmallows. For an object to be bouyant, or float, it must have less density that what it is floating in, or it has to have something attached to it that helps it float – like you with a life jacket on. Here is another image of a density tower I made. As you can see, a ping pong ball, wooden cube, and foam cube all floated, meaning their density was lower than the density of the rubbing alcohol. The die, screw, and penny all sunk to the bottom, meaning they had a high density, at least equal or higher to that of the honey at the bottom.
This explanation from stevespanglerscience.com is an excellent description of all of the learning possible in this experiment. "Density is basically how much “stuff” is packed into a particular volume. It’s a comparison between an object’s mass and its volume. Remember the all-important equation: density = mass ÷ volume. Based on this equation, if the weight (or mass) of something increases but the volume stays the same, then density goes up. Likewise, if the mass decreases but the volume stays the same, then density goes down. Lighter liquids (like water or vegetable oil) are less dense than heavier liquids (like honey or corn syrup) so they float on top of the heavier liquids. The same amount of two different liquids you used in the container will have different densities because they have different masses. The liquids that weigh more (a higher density) will sink below the liquids that weigh less (a lower density)." -https://www.stevespanglerscience.com/lab/experiments/density-tower-magic-with-science/ Some of the items appeared to get "stuck" in a layer. If an object comes to rest in a certain layer, it actually means that it is less dense than the layer below it and more dense than those above it. Well, that's all for now. Hope this post helped! We acted as lawyers last week and presented cases in fractions court. The kids read their case file, chose a side, and then presented their case. They did a great job. Here are some pictures.
We started working on subtracting mixed numbers this week and in order to model the process, made mini sheet cakes, brownies, pizzas, etc. to cut up and share. Here are the kids creating their culinary masterpieces. And speaking of adding and subtracting fractions, I posted some videos I made on my math blog to help you! Here is the link: https://wippich.weebly.com/math-blog/videos-to-help-with-adding-and-subtracting-fractions
|
Archives
June 2019
Categories
All
|




























 RSS Feed
RSS Feed