|
In class we've been talking about matter and how all matter is made up of atoms. Matter can be made up of:
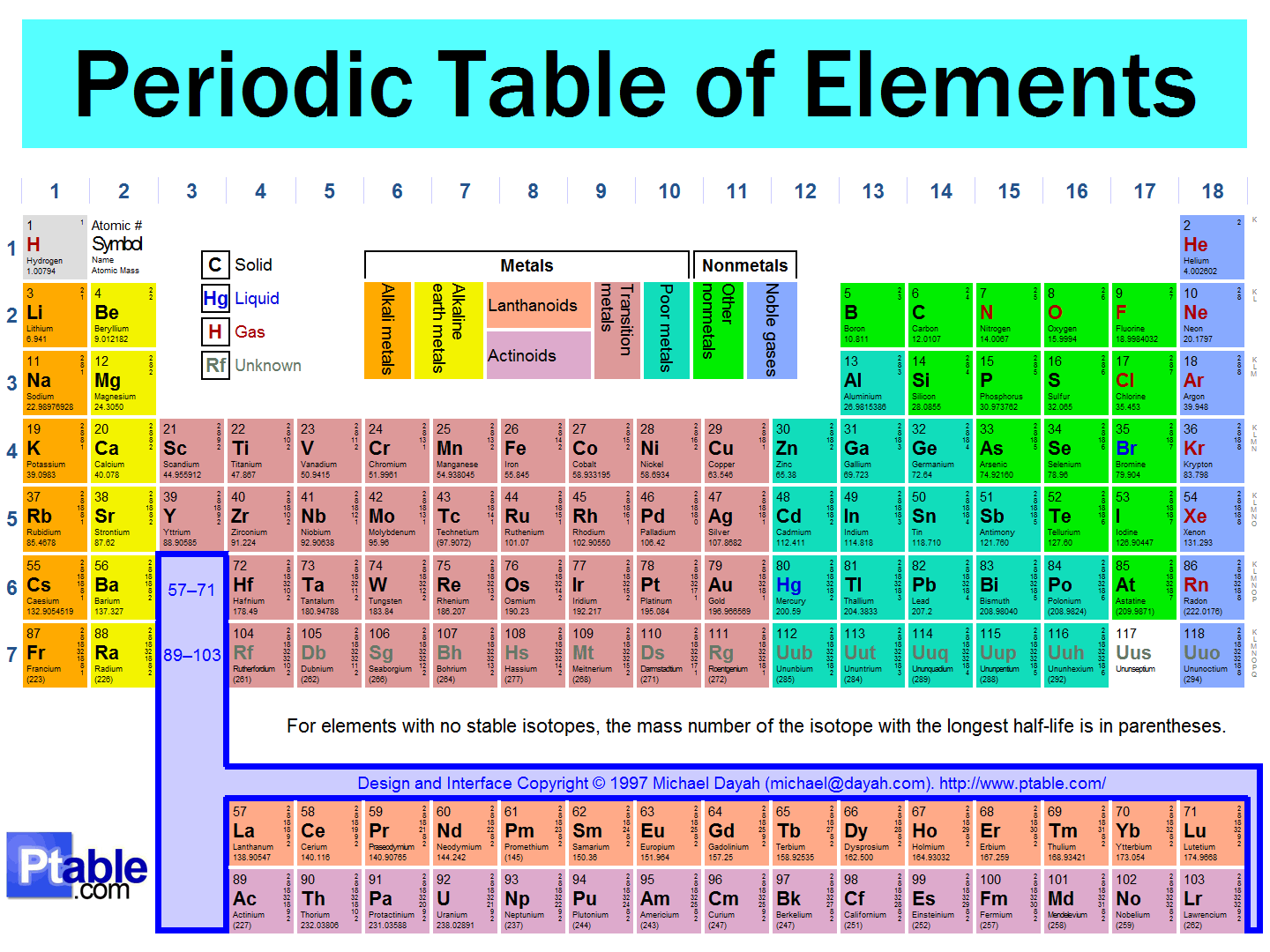
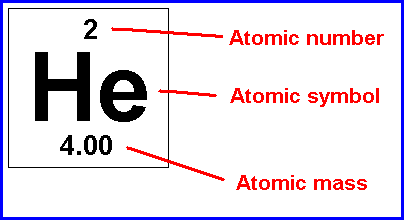
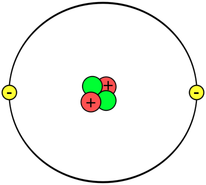
There are 118 known elements (some naturally-occurring and some man-made). Their names and properties can be found in the PERIODIC TABLE of ELEMENTS, as shown below. You do NOT need to memorize all of the elements for fifth grade! We then learned that those atoms are made up of things as well. The kids asked the question, "If atoms are the smallest pieces of matter, then how can atoms be made up of "stuff" if atoms are the smallest units of "stuff"???" Read on to find out :) Well, the simple answer is that these small bits of the atom- protons, neutrons, and electrons (also known as sub-atomic particles)- cannot exist on their own. They HAVE to co-exist with the other sub-atomic particles. And yes, these sub-atomic particles are made of even SMALLER things (quarks, etc.) that also can't exist on their own, but you can find out more about that in your own research or future physics classes ;) Anywho, I digress. In class before break we constructed our own atom models using clay, pipe cleaners, and pointy wooden rods of science (okay, toothpicks). Below you can see the periodic "block" for Helium. We talked about how this "block" shows us a lot of information about the element helium and what an atom of helium looks like. First, we talked about how the atomic number at the top of the box shows us the number of PROTONS in the nucleus, or the part in the center, of the atom. These protons have a POSITIVE charge (remember the two Ps go together: proton=positive). Also in the nucleus of an atom are NEUTRONS (remember Nucleus has Neutrons) and these subatomic particles have a NEUTRAL (not positive or negative), or NO charge. We'll talk more about how to find the number of neutrons in a moment after we discuss the third subatomic particle. Far outside of the nucleus (remember, the nucleus=protons and neutrons bundled together in the center) are ELECTRONS. Electrons have a negative charge. How many electrons are in an atom? The same number as there are protons! Scientists used to think these electrons circled the nucleus like planets orbit the sun, but now we know that their motions are a bit more like a moth buzzing around a light at night. Below is an example of how drawings of atoms are usually depicted. The picture is a model of a helium atom with 2 protons, 2 neutrons, and 2 electrons. You can differentiate between the subatomic particles in the image by their charges: (+ = proton, - = electrons, no charge = neutrons). So how do we figure out the number of neutrons in an atom? Well, you'll notice that each element in the periodic table has two numbers in the box- one at the top (the atomic number, which is the number of protons in the atom) and one at the bottom which is the average atomic weight (it is listed as atomic mass above). To find the number of neutrons: 1. Round the ATOMIC WEIGHT to the nearest whole 2. Subtract the ATOMIC NUMBER (number of protons) from the average ATOMIC WEIGHT (see example below). For Lithium (in image below), we find that the atomic weight is 7, the atomic number is 3. 7 - 3= 4 neutrons 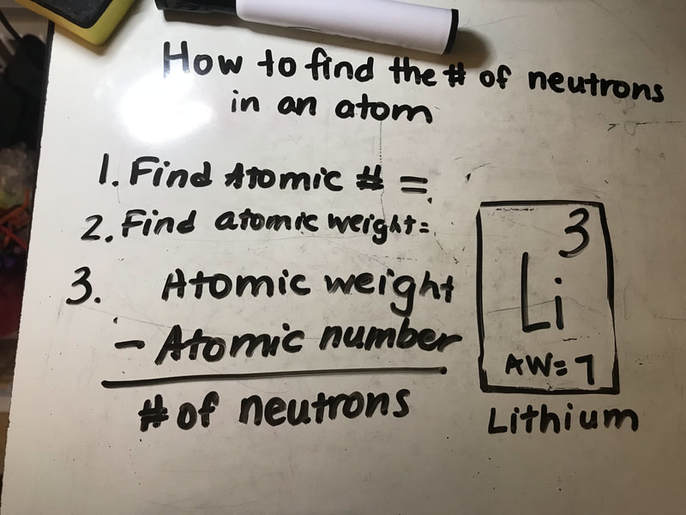 How to find the number of neutrons How to find the number of neutrons And here are pictures of a couple of our Helium atoms and the kids making them :) Now, let's talk about the size of subatomic particles in comparison to atoms. Imagine the atom is the size of a professional football stadium. In the center of the field, right in the middle of the 50 yard line, would be the nucleus. How big would the nucleus be in comparison to the stadium? The size of a GRAPE! The nucleus is SUPER small compared to the size of the atom as a whole.
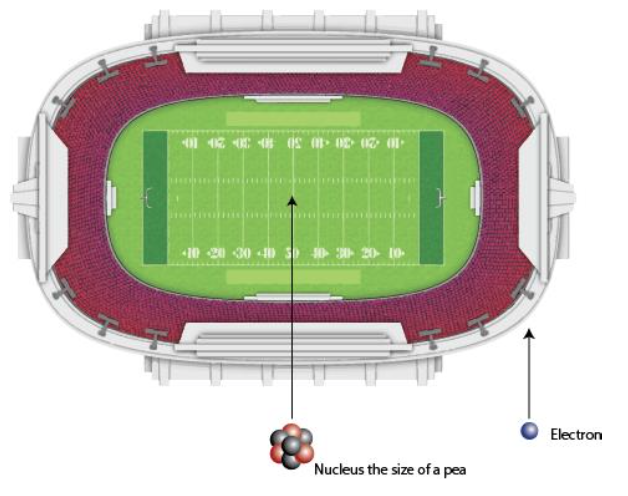
Now, we know that electrons buzz around the nucleus. In our football stadium atom, these electrons would each be the size of a grain of sand in the VERY last row. Imagine all of that empty space in between that grape in center-field and those grains of sand in the last row. Pretty amazing, right? Atoms are made up of mostly empty space! Here is an image I found online to help you "see" this better, however, in this example they say the nucleus is the size of a pea!
0 Comments
Last week we used Legos to learn about atoms, molecules, elements, and compounds! First, we discussed that atoms are the basic building blocks of matter- every object is made of atoms! We talked about how small atoms are compared to things and "zoomed in" to see an atom up close using the cool website below. learn.genetics.utah.edu/content/cells/scale/ Then, we talked each of the four terms.
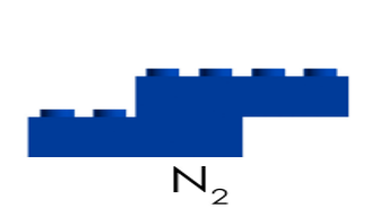
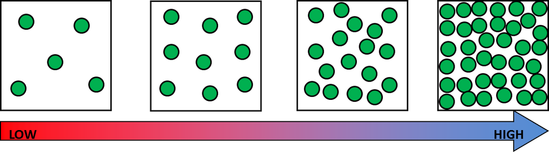
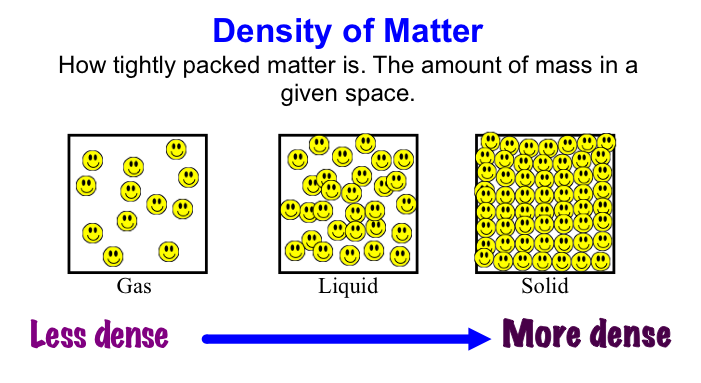
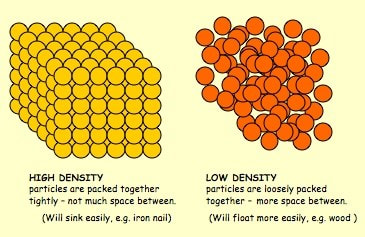
Next we made some atoms and molecules! Here is an example of the activity we did. First I ask the students to take one lego and hold it up. This is considered one atom. This is one atom of nitrogen, for example. We then discuss how this is not only an atom, but it is also an example of an element because it is a pure substance- there are no other types of atoms bonded to it. Next, we find another identical atom to the first (same color and shape) and bond it (connect) to the first one. We talked about how this is a molecule now because it is 2 or more atoms bonded together. It is also still considered an element because it still has only one type of atom in it, nitrogen. We put the little 2 below the N to let us know that it has two atoms of nitrogen. No matter how many atoms are in a molecule, 1 or 100, if they are all the same kind of atom, they are considered a molecule of that element. Next, we found two different colored Legos and bonded them together. We called this a molecule, because it is two or more atoms bonded together (molecules can be same or different, in this case they are different) and we also called it a compound because it is a molecule with two or more types of atoms. In this case we have a sodium (Na) atom and a chlorine (Cl) atom combining to make NaCl, sodium chloride, or salt! We talked about how this is not an atom because it is not just one thing and it is not an element because it is not a pure substance only made of one type of atom. We've done a couple of demonstrations in class the last couple of days to discuss the difference between volume, mass, and density. First we reviewed what "matter" is- anything that has mass and volume. Some people think that matter makes up "everything" but that is not accurate because matter does not make up thoughts, feelings, ideas, actions, etc. It DOES, however, make up every object. Next we reviewed the term "volume" as being the amount of space something takes up. I showed the kids three balls and we set out to answer the following question: How do the masses and volumes of a baseball, tennis ball, and mini basketball differ from each other? Like in the pictures below, the balls all had the same volume- they took up the same amount of space. Then we used a balance scale to find the mass of 3 balls and compared the masses. I apologize for the length of these videos- I don't have time to edit them this evening. Our results looked like this: Basketball rubber ball- 18g Tennis ball- 58 g Baseball- 148g Then we discussed the differences between each ball's mass versus its volume. The baseball had a lot more mass in the same amount of space than the other two balls. This means that it has a higher density, its molecules are packed much more tightly together. We will learn more about molecules later in the unit. Here are some graphic representations of density. As for floating (buoyancy), it isn't just how "heavy" something is that determines if it will sink or float, but a lot of it has to do with density. This explanation on density and buoyancy from http://www.indypl.org/kids/blog/?p=9042, the The Indianapolis Library's website, is excellent: Every object on earth is made of atoms. Gravity pulls these atoms to the earth. You can measure the pull of gravity on an object – we call that measurement weight. Density is how close together the molecules of a substance are or how much mass a substance has in a given space. If you have one cup of jelly beans and one cup of marshmallows…the jelly beans have more mass…there is more “stuff” compacted into the cup. The marshmallows are mostly air. If you put each of those cups in a microwave to melt…the sugar and water that makes up the jelly beans would almost fill the cup to the top. The sugar and water that makes up the marshmallows would only fill the cup a little bit because marshmallows have less mass, they are mostly made of air. Materials with more density weigh more. A cup of jelly beans weighs more than a cup of marshmallows. Today we explored more with density by making a density tower. We poured 100 ml of various liquids into a large graduated cylinder and watched as the liquids separated into different layers. Then we added some objects to the liquid to see what would happen. Here are a few photos of the tower: Unfortunately I only had light cream instead of whole milk so our water and milk blended together and look like one big layer. Here is the order of the liquids: As you can see, a ping pong ball, wooden cube, and foam cube all floated, meaning their density was lower than the density of the rubbing alcohol. The die, screw, and penny all sunk to the bottom, meaning they had a high density, at least equal or higher to that of the honey at the bottom.
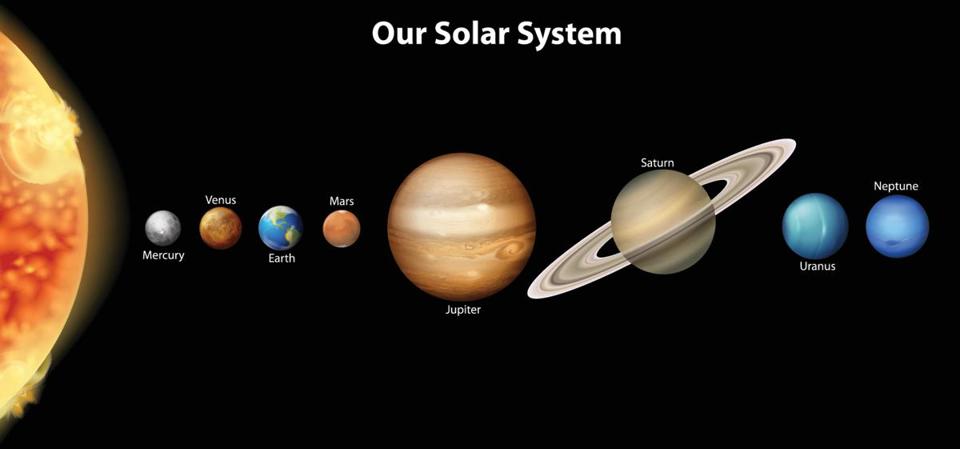
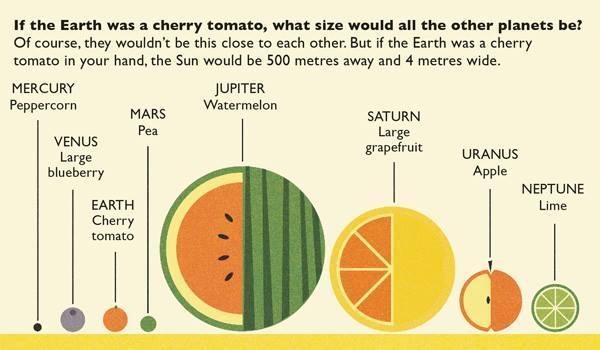
This explanation from stevespanglerscience.com is an excellent description of all of the learning possible in this experiment. "Density is basically how much “stuff” is packed into a particular volume. It’s a comparison between an object’s mass and its volume. Remember the all-important equation: density = mass ÷ volume. Based on this equation, if the weight (or mass) of something increases but the volume stays the same, then density goes up. Likewise, if the mass decreases but the volume stays the same, then density goes down. Lighter liquids (like water or vegetable oil) are less dense than heavier liquids (like honey or corn syrup) so they float on top of the heavier liquids. The same amount of two different liquids you used in the container will have different densities because they have different masses. The liquids that weigh more (a higher density) will sink below the liquids that weigh less (a lower density)." -https://www.stevespanglerscience.com/lab/experiments/density-tower-magic-with-science/ Some of the items appeared to get "stuck" in a layer. If an object comes to rest in a certain layer, it actually means that it is less dense than the layer below it and more dense than those above it. Well, that's all for now. Hope this post helped! Today we started on our new unit in science- MATTER! In order to kick off the unit, I did a demonstration to get the kids excited about the topic. Since a lot of people assume that gases have no mass, I wanted to challenge their thought processes. Here is a video of the demo: After the video, I explained how the demonstration worked. Contrary to popular belief, gases DO have mass and when I added the baking soda to the vinegar, a chemical reaction happened that produced the gas carbon dioxide has more mass than the air we breathe. As I poured the invisible gas into the bag, it caused the balance scale to tip to the side with the gas with the higher mass. After discussing the scale activity, we turned to some very important vocabulary work that we will focus on during this unit. We were introduced to the following words: Matter- anything that has mass and takes up space Mass- the amount of matter in an object; in science we measure in kilograms, grams, etc. Volume- the amount of space an object takes up Density- the amount of mass packed into a given space (volume) Weight- a measure of the gravitational pull on an object; a force we measure in Newtons A student asked a very interesting question about the volume of objects. He wondered if we were to take a pencil and mold the same stuff into a new shape if it would have the same volume. To explore this, I modeled a couple of different shapes that I made with a ball of clay. First I made it into a ball and let everyone look at it. I then rolled it into a long snake-like shape and asked the kids which one had a greater volume. They realized that because they both were made using the same amount of clay, they had the same volume even if they looked different. We then practiced this in a hands-on way by using Minecraft cubes. The kids were given the task to make a shape using 6 cubes. We had a variety of different shapes. We looked at our creations (see below) on the Promethean and discussed how they all had the same volume because they were all made with exactly 6 cubes. We will get into volume and its formula later in the year. We then briefly talked about the term density. We will be revisiting all of these terms over the coming days and weeks so no worries if you don't get it yet! When talking about weight, we reviewed that gravity is a force that pulls objects toward one another. We discussed how we are pulled toward the center of Earth which is why we don't go flying off into space. We also talked about how the sun's gravitational pull keeps all of the planets in orbit around it. We also talked about how moons orbit their planets due to each planet's gravitational pull. We then had a brief refresher of fourth grade science and discussed the planets and their order from the sun. We discussed mnemonics to help us remember the order of the planets: My (Mercury) Very (Venus) Educated (Earth) Mother (Mars) Just (Jupiter) Served (Saturn) Us (Uranus) Nachos (Neptune). After this discussion, we reviewed the order of the planets according to size, also a fourth grade SOL. We did this by comparing them to food :) Then we talked about how our weight would be different if we were to go to another planet, or even the moon. We'd still have the same MASS (all of our body would have the same amount of stuff in it), but the different gravitational pulls of the planets would have different effects on our mass, thus making us weight different amounts on each planet.
For example, if a child with a weight of 30kg on Earth were weighed on Mars (which has a gravitational pull that is only about 2/5 of Earth's), they would only weigh 11.3 kg on Mars. They still have the same amount of stuff (mass) in them, their mass just isn't being pulled toward the center of Mars as much as it is pulled toward the center of Earth. On Jupiter, that same 30kg child would way 75.8kg! The amount of gravitational pull a planet or other object has depends on its mass- the larger the object's mass, the stronger its gravitational pull. After these discussions, worked on calculating our weights on the different planets and Earth's moon. Here is a website that will let you do the same thing! Very cool! www.exploratorium.edu/ronh/weight/ More on these topics later :) |
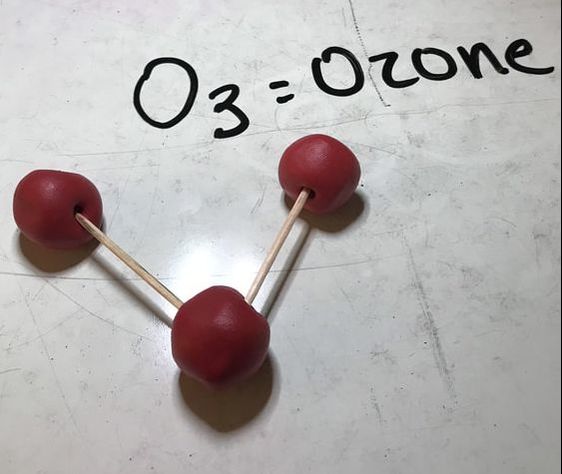
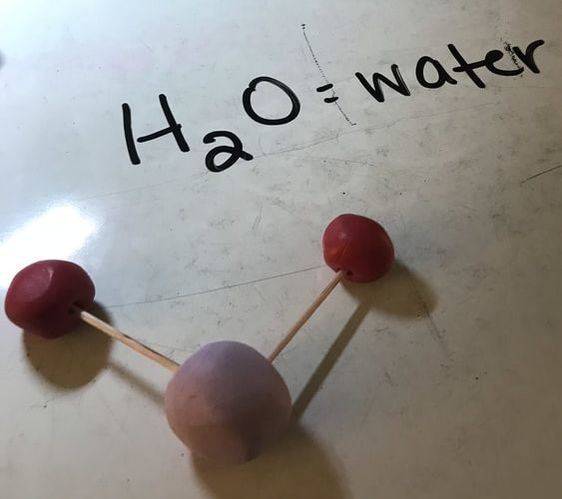

































 RSS Feed
RSS Feed